 They found from their observations that the majority of alpha particles passed through the gold foil as expected. When they fired the alpha particles at the screen, Geiger and Marsden recorded any flashes which occurred. The zinc sulphide screen emits flashes of light known as scintillations when it is hit by alpha particles. The set-up for Geiger and Marsden’s experiment is shown in the diagram below: If Thompson’s model of atomic structure was correct, the alpha particles should either pass straight through the gold foil or be only slightly deflected. They fired alpha particles at a very thin layer of gold foil, one atom in depth. These two scientists were Hans Geiger and Ernest Marsden, and they had been conducting experiments looking at the behaviour of alpha particles.Īlpha particles will be explained in more detail later in the chapter but in brief they can be described as helium nuclei and are uncharged particles. In the early 20 th century, the scientist Ernest Rutherford discovered the presence of the nucleus of the atom by looking at the findings from experiments conducted by two scientists working in his laboratory. This is why the model became known as the plum pudding model. Thompson thought that an atom looked like a ball of positive charge with electrons dotted through it, like plums in a pudding. Thompson had discovered the first of the sub-atomic particles: the electron. Thompson discovered that atomic structure was actually not a hard sphere but in fact contained smaller negatively charged particles which had very little mass. Dalton believed that atoms of an element have the same mass and that each different element had its own type of atom with different masses. 
These substances we now know as elements. Dalton claimed that some substances were made up of identical atoms. 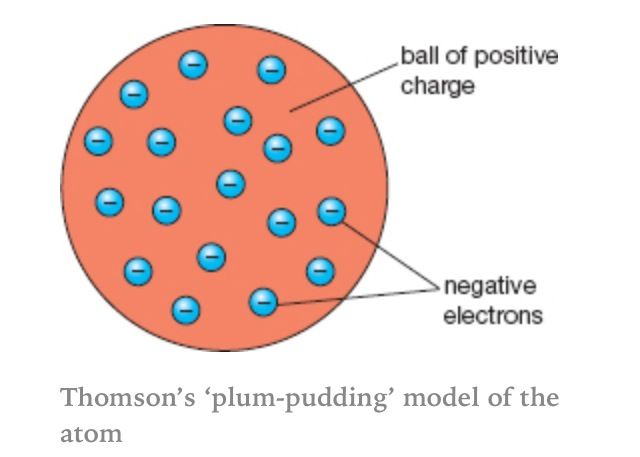
In 1830, John Dalton – a scientist who studied gases in the 19 th century – stated that all atomic structure was made up of atoms which he believed to be tiny, hard spheres. The work of many scientists over the years has contributed to the model of the atom we use today. 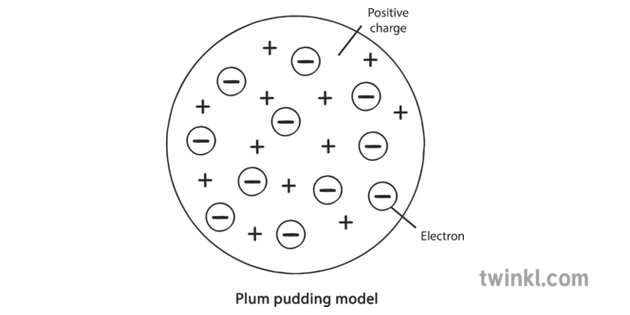
The model of the atom has been developed over the years as advances have been made in scientific methods and technology. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
